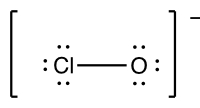The formal charge is the number of valence electrons in a neutral atom minus the number of electrons the atom owns in the compound. Formal charges provide a way for one to determine the most reasonable or best Lewis structure as well as the more likely structure if we are not sure how the atoms are connected.
# valence e– in neutral atom – the # e– owned by the atom in the molecule
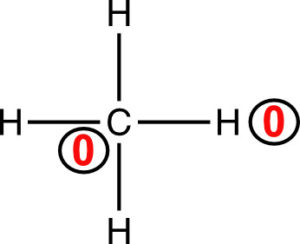
An atom in a molecule owns half of its bonding electrons and it owns its lone pairs (or even a single electron as in a free radical). Let’s look at the formal charges for the atoms in methane. First, draw the Lewis structure.
Then count the number of lone pairs on the carbon atom. There are 0 lone pairs on carbon. Then count the number of bonding electrons. Carbon has 8 bonding electrons. One-half of 8 is equal to 4 electrons. Carbon owns 4 electrons in the compound. The number of valence electrons for the neutral carbon atom is 4.
The formal charge on carbon is 0. The hydrogens each own 1 electron, and
Both carbon and each of the 4 hydrogens in methane have a formal charge of zero. The formal charges are written next to the atom and circled.
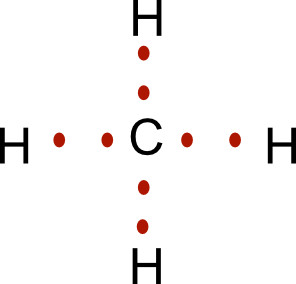
Another way to do this is to draw the Lewis structure and replace the single bonds with the bonding electrons.
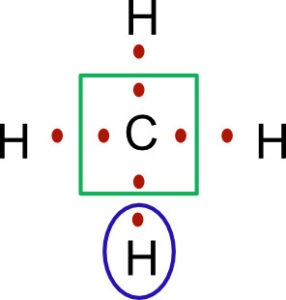
Circle the electrons around the carbon. We can see the carbon owns 4 electrons. Circle the electrons around hydrogen, and we see each of the hydrogens own one electron.
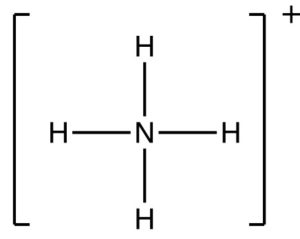
We can take a look at NH4+ ion. First draw the Lewis structure, and then count the number of electrons owned by nitrogen in the compound.
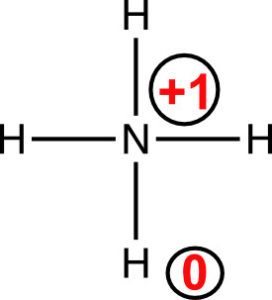
The neutral nitrogen atom has 5 valence electrons, and the atom in the compound owns 4 electrons.
The nitrogen has a formal charge of +1, and the hydrogen atoms have a formal charge equal to zero.
There are times when one needs to determine a more important resonance structure or determine how the atoms are connected in some compounds. Here are the rules for using formal charge to choose the best Lewis structure.
2. The same nonzero formal charges on adjacent atoms are not preferred.
3. A more negative formal charge should be on the more electronegative atom.
4. A more positive formal charge should be on the less electronegative atom.
Take a look at the following structures. We need to determine which is the most reasonable Lewis structure fo the OCN– ion.
First, determine the formal charges on all of the atoms in the three structures. The formal charges must add to the charge on the species. If it is a neutral compound, formal charges must add to zero. If it is an ion, the formal charges must add to the charge on the ion.
The first structure is the most reasonable structure. The formal charges add to -1 and the more negative formal charge is on the more electronegative element. The third structure has a -2 charge on the nitrogen atom and the second structure has a -1 on the nitrogen atom which is less electronegative than oxygen.
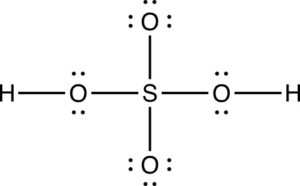
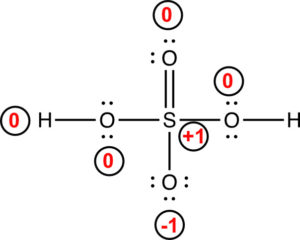
The Lewis structure for H2SO4 is below, but is it the best Lewis structure?
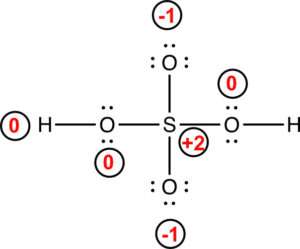
The formal charges for each of the atoms are indicated on the figure below.
The structure is valid, but it can be improved. There is a formal charge of +2 on the sulfur. If we move a pair of electrons from oxygen to form a double bond with sulfur, we end up with the following Lewis structure.
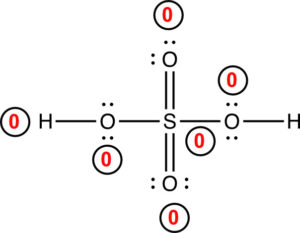
Now, there is a +1 formal charge on sulfur. We can move another pair of electrons from the other oxygen to form another double bond. The structure is:
The structure above is the best Lewis structure. All of the atoms have a formal charge of zero. For compounds try to draw the structure so the formal charges on each atom is equal to zero. In some cases, this is not possible. One example is for CO in the exercises. Recall, that carbon must always have an octet of electrons.
Exercises
Exercise 1. What are the formal charges on the oxygen and chlorine in the following structure?
Exercise 2. Use formal charges to determine the best Lewis structure for carbon monoxide, CO.
Exercise 3. Draw resonance structures for NOCl. Use Lewis structures to determine the more important resonance structure.
Exercise 4. Use formal charges to draw the best Lewis structure for SCN–
Exercise 5. Use formal charges to draw the best Lewis structure for H3PO4
Check Solutions/Answers to Exercises
Back to Covalent Bonds and Lewis Dot Structures
Back to General Chemistry 1 Study Guides
Back to Home Page