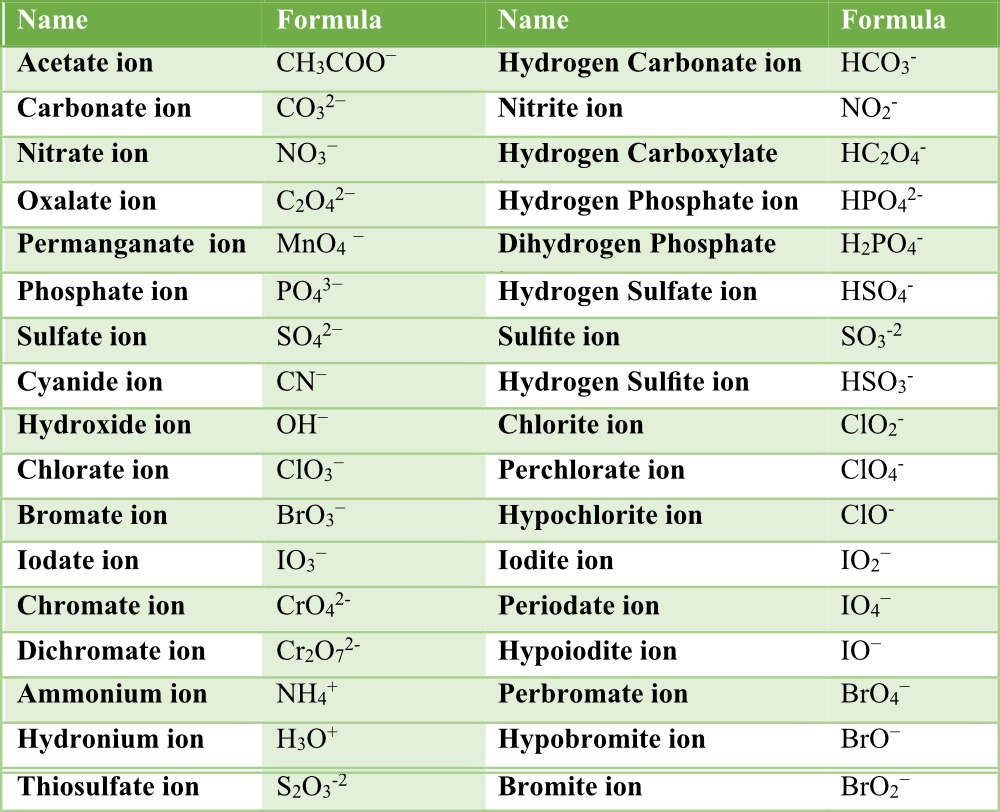
In this study guide we will discuss polyatomic ions and how to name ionic compounds with polyatomic ions. First, let’s define a polyatomic ion. A polyatomic ion is a group of atoms covalently bonded together with a charge that affects the whole polyatomic unit. Some examples of polyatomic ions are in the following table. Make sure you memorize the names, formulas, and charges of each of the ions in this table.
Note, most of the polyatomic ions are negatively charged with the exception of NH4+ and H3O+. Once you have memorized these ions, you will be able to name many different compounds including acids and bases. If you memorize the first column of ions, you can derive the rest of the ions. For example, NO3– is nitrate ion. Looking at NO2– we see it is nitrite ion. The nitrite ion has one less oxygen than the nitrate ion, but the charges are the same. Sulfate ion, SO42-, has 4 oxygens and a charge of -2. The sulfite ion, SO32-, has one less oxygen, but the same charge.
Now we can take a look at phosphate ion, PO43-. Adding a proton
gives hydrogen phosphate ion, HPO42-. Add a proton to hydrogen phosphate
and dihydrogen phophate, H2PO4–, is formed. Finally, add a hydrogen to H2PO4–
and phosphoric acid is formed. The acid must be neutral before naming it with the “ous” or “ic” suffix. More about naming acids in the next study guide.
As when naming simple ionic compounds, the name of the metal ion comes first and is followed by the name of the polyatomic ion. For example, sodium (Na+) and sulfate ion (SO42-) would give Na2SO4, sodium sulfate. Recall, the compound is neutral and the charges must add to zero.
For compounds that contain more than one polyatomic unit, use parenthesis. If we combine ammonium ion, (NH4+) and carbonate ion (CO32-) we have ammonium carbonate, (NH4)2CO3. Two ammonium ions are required for each carbonate ion.
When the metal cation can have more than one charge, use Roman numerals to indicate the charge. For example, chromium ions, (Cr3+) and nitrate ions (NO3–) would result in chromium (III) nitrate, Cr(NO3)3, whereas, chromium (VI) ions and nitrate ions would combine to form chromium (VI) nitrate, Cr(NO3)6.
Note the halides, I, Cl, and Br, when combined with oxygen, can form four polyatomic ions. For Chlorine, the two additional ions are perchlorate ion, with four oxygens, and hypochlorite ion with only one oxygen. Please refer to the table for the names and formulas of the ions.
Exercises
Exercise 1. Name the following compounds
1. Fe(OH)3
2. Mg3(PO4)2
3. CuNO3
4. Ca(CH3COOH)2
5. Co(HCO3)2
Exercise 2. Write chemical formulas for the following.
1. copper (II) sulfate
2. strontium hypoiodite
3. manganese (IV) perbromate
4. iron(II) phosphate
5. ammonium hydrogen sulfite
6. barium chlorate
View solutions to exercises
Back to Study Guides for General Chemistry 1
Back to Home Page