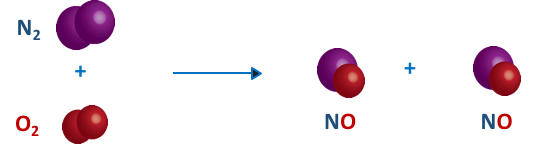
Now that we have defined an element as a collection of just one type of atom, we shall now consider the second type of pure substance, the compound. Unlike an element, a compound is a collection of more than one type of atom. Recall that both elemental nitrogen (N2) and elemental oxygen (O2) occur as diatomic species. However, elemental nitrogen and elemental oxygen can be mixed together under certain conditions to form a new species in which one oxygen atom is bonded to one nitrogen atom as shown in the figure. This new species is called a compound. By definition, compounds are pure substances consisting of more than one type of atom. As can be seen in the above reaction, a new chemical substance is formed, nitric oxide, NO, a compound composed of both one nitrogen atom and one oxygen atom.
Some common examples of compounds are carbon monoxide (CO), sodium bicarbonate (NaHCO3), dihydrogen monoxide (H2O), sodium chloride (NaCl), and calcium carbonate (CaCO3). The symbolic notations, NO, CO, and NaHCO3, are chemical formulas. Chemical formulas indicate the type and number of each atom in the compound. The subscripts indicate the number of atoms in the compound. For example, Na2CO3 is composed of 2 sodium atoms, 1 carbon atom, and 3 oxygen atoms. 
The compound nitric oxide has the chemical formula NO. This compound contains 1 nitrogen atom and 1 oxygen atom. Water has the chemical formula H2O and contains 2 hydrogens and 1 oxygen. Elemental oxygen, O2 contains two oxygen atoms. Baking soda (sodium bicarbonate), NaHCO3 has 1 sodium atom, 1 hydrogen atom, one carbon atom, and 3 oxygen atoms.
Compounds are also defined as having constant composition. That is, the compound carbon monoxide, CO, must always consist of one carbon atom and one oxygen atom. To add an additional oxygen atom to this compound would yield a new substance, carbon dioxide (CO2). By the same token, to add a second oxygen to water, H2O, would yield hydrogen peroxide, H2O2.
The atoms in a compound are chemically combined. Compounds can be chemically broken down into their elemental components by chemical means. For example, hydrogen peroxide, H2O2, can be broken down into elemental hydrogen and elemental oxygen. Hydrogen and oxygen gas can be produced from water, H2O, when an electrical current is passed through it. Recall that unlike compounds, elements cannot be chemically broken down into simpler substances.
In summary,
A compound is a pure substance.
Compounds contain two or more different elements that are combined.
Chemical formulas indicate the type and number of each atom in a compound.
Compounds have constant composition.
Compounds can be chemically broken down into simpler substances by chemical means.
Click Here for Compounds Worksheet
Click Here for Elements, Compounds, and Mixtures Worksheet
Exercises for Compounds
1. Write the chemical formula for caffeine, which has 8 carbon atoms, 10 hydrogen atoms, 4 nitrogen atoms, and 2 oxygen atoms.
Check Answer and/or View Worked out Solution to Exercise 1
2. Write the chemical formula for propanol, which has 3 carbons, 7 hydrogens, and 1 oxygen.
Check Answer and/or View Worked out Solution to Exercise 2.
3. Phenylanaline is an amino acid that has the chemical formula C9H11N2O2. Name the elements in phenylalanine. What is the total number of atoms in the formula for phenylalanine?
Check Answer and/or View Worked out Solution to Exercise 3.
4. Hydrogen peroxide has the chemical formula H2O2. It breaks down to form liquid water, H2O and oxygen gas, O2. Identify which substances are elements and which are compounds.
Check Answer/Solution to Exercise 4
Back to Ions, Atoms, and Molecules
Back to Study Guide List for General Chemistry 1
