Matter can be classified into two separate groups. The first group are pure substances which consist of elements and compounds. An element is a pure substance that cannot be chemically broken down into simpler substances. Elements are composed of atoms. An atom is the smallest particle of an element that still retains all of the element’s characteristics. A compound is a pure substance that can be broken down chemically into simpler substances. The second group consists of mixtures of pure substances and may be considered an extension of the first group.
In this module, we will classify elements and their properties using the periodic table. In addition, the Law of Mass Conservation will be used to determine the mass of products formed in a chemical reaction. The Law of Multiple Proportions will be discussed. Thomson’s cathode-ray experiment, Millikan’s oil drop experiment, and Rutherford’s gold foil experiment will be explained in terms of how they contributed to modern atomic theory.
The structure and size of atoms will be discussed and the number of atoms in a given sample will be calculated. Atomic mass will be calculated when give the fractional abundance (or percent abundance) and mass of each isotope. We will classify bonds as ionic or covalent and classify matter as a mixture or pure substance. Conversions between grams and numbers of moles or atoms using molar mass and Avogadro’s number will be shown. Finally, nomenclature will be presented.
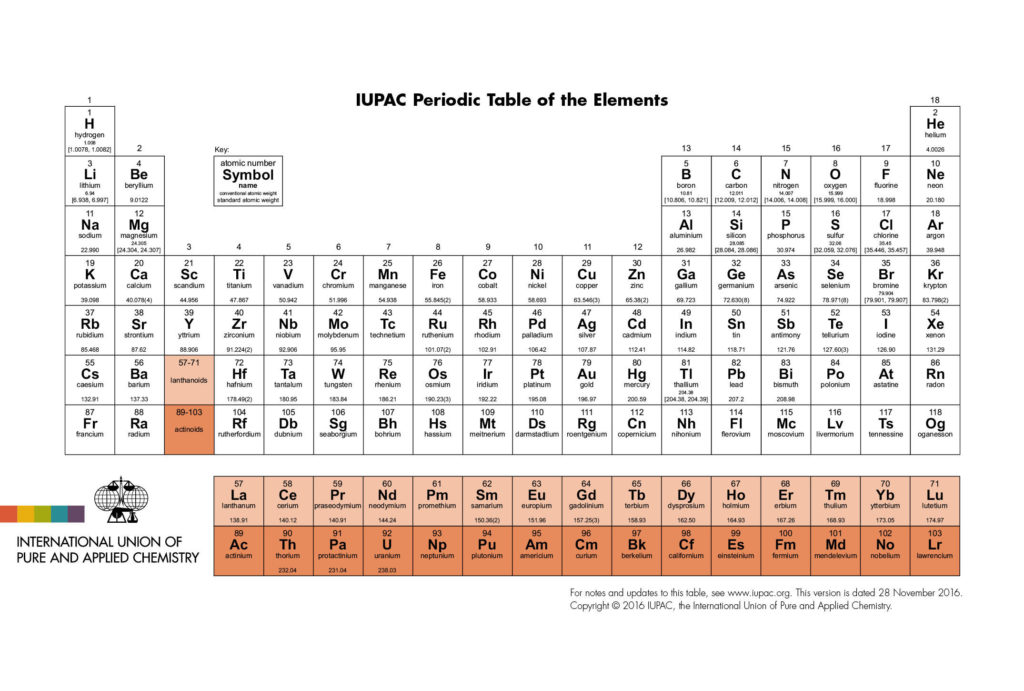
There are 118 known elements in the universe. Most of us are already familiar with some of their names. We are well acquainted with the names carbon, gold, copper, iron, oxygen, and hydrogen to name a few. These are all elements, each of which is made up of its own unique type of atom. An atom is the smallest particle of an element that still retains all of the characteristics of that element. The Periodic Table lists the names and symbols of all the known elements. Below, is the updated periodic table which lists the names of elements 113, 115, 117, and 118. The periodic table in your textbook most likely does not include the latest updated periodic table. For more information on the naming of elements please go to The International Union of Pure and Applied Chemistry, IUPAC.org. Exploration for new elements beyond the 7th period continues. Detailed information on the requirements for new elements and how elements are named can be found at IUPAC.org. Click on the periodic table to see it full size.
Each element has its own position on the periodic table. The element hydrogen is located in the upper left-hand corner of the periodic table. The elements are referred to by one and two letter symbols. One letter symbols are always capitalized while only the first letter of a two letter symbol is capitalized and the second letter is lowercase. The element symbol for hydrogen is the capital letter H and the element symbol for sodium is Na. There is a number directly above each element symbol. This number is called the atomic number and is represented by an uppercase Z. The atomic number for hydrogen is 1. The element symbol for gold is Au, and the atomic number is 79. The elements are arranged on the periodic table by atomic number. It is the atomic number that is used to identify an element.
At the beginning of their chemistry studies, many chemistry students confuse the terms atoms, elements, and elemental. Are they the same in essence? Are the terms interchangeable? You can think of an element as the name of a certain type of atom. For example the element carbon is made up of carbon atoms, and the element nitrogen is made up of nitrogen atoms. The term elemental is used to describe the most stable physical state that an element exists in under normal conditions–normal temperatures and pressures. For example, the most stable form for chlorine atoms is as discrete diatomic structures, i.e. two chlorine atoms bonded together comprise one unit or molecule of elemental chlorine.
On the periodic table, the symbol for chlorine is Cl, the monoatomic species. When referring to the elemental form of chlorine, the chemical symbol is Cl2, the diatomic species. Elemental chlorine occurs as a gas in nature. 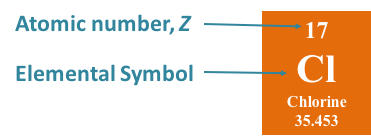
In contrast to elemental chlorine, carbon atoms in the most stable state occur in the form of solid graphite. Pencil lead is an example. This form of carbon occurs as six-membered rings that are fused together producing extensive sheets which form layers. Even though this structure of elemental carbon seems complex, its chemical symbol is a single C.
Regardless of how atoms bond together or whether they exist as a gas, liquid, or a solid in an elemental state, the primary difference between different elements is the type of atoms involved. For example, the element carbon is made up of carbon atoms, whereas the element gold is made of gold atoms. We will address this difference in detail later, but for now understand that any atom can be broken down into a collection of its subatomic building blocks–protons, electrons, and neutrons which will be discussed in more detail later–by doing so, the atom would lose its elemental identity, and that both the carbon and gold atoms would become just piles of subatomic particles.
Remember, when talking about elements, we are referring to those atoms in their most stable form. That is, elemental sodium occurs as a collection of individual sodium atoms. Elemental sodium is monotomic whereas elemental oxygen occurs as a collection of two oxygen atoms bonded together. Oxygen is a diatomic species. Phosporous, in its most stable elemental form, will occur in groupings of four phosphorus atoms. The symbol for elemental phosphorus is P4. This is called a tetratomic species.
Click Here for Elements, Compounds, and Mixtures Worksheet
Back to Atoms, Molecules, and Ions
Study Guide List for General Chemistry 1