First we will learn how to name binary covalent compounds. The smallest discete unit of a covalent compound is the molecule. In naming binary covalent compounds, compounds that are composed of two different nonmetal atoms, the prefixes in the table are used to indicate the number of each atom in the compound. These prefixes must be memorized.
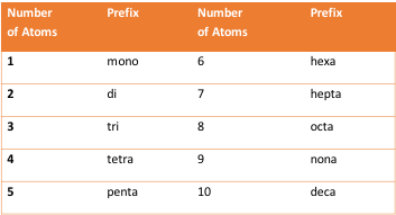
There are three rules in naming binary covalent compounds:
1. Never use the prefix mono- in naming the first atom of a covalent compound.
2. If an atom in the compound starts with a vowel, drop the vowel at the end of the prefix.
3. Modify the suffix of the second atom to have an –ide ending.
In naming the compound CO, all three rules are demonstrated.
CO is called carbon monoxide. The prefix mono- is used for the second atom but not for the first atom. The second atom, oxygen, becomes oxide. Because oxygen begins with a vowel we drop the “o” at the end of mono-. We have mono + oxide = monoxide.
NO2 is named nitrogen dioxide, SCl2 is named sulfur dichloride, and CCl4 is named carbon tetrachloride.
What about CH4? Is it named carbon tetrahydride? No, this compound is named methane. It is just one of many hydrocarbons, compounds with only carbon and hydrogen atoms. Methane is classified as an alkane, a hydrocarbon where carbon is bonded to four atoms (hydrogen or carbon). You will learn more about the many alkanes in organic chemistry, but for now it is important that you memorize the names and formulas of the first 10 alkanes. Most organic nomenclature is based on these alkane names.

Exercises
Exercise 1. Write a chemical formula for each the following:
a) dinitrogen pentoxide
b) iodine tetrachloride
c) sulfur hexafluoride
Exercise 2. Name the following:
a) CS2
b) C8H20
c) P2O5
