Intramolecular or bonding forces are found within a molecule. The chemical behavior of each phase of matter is the same because the same particle is present whether the substance is a liquid, solid, or a gas. For example, H2O molecules are present in the solid, liquid, and gas phase. Intermolecular or nonbonding forces are found between molecules. The strength of these attractive forces in each phase of matter is different because the strength of the forces differ between phases. We refer to intermolecular forces as van der Waals forces. We will discuss three types of intermolecular forces: London dispersion forces, dipole-dipole, and hydrogen bonding.
Here, we examine both the kinetic energy and the properties of the three different phases of matter.
The particles of a liquid have strong attractive forces, but not as strong as in the solid phase. A liquid is fluid, therefore, the particles can slide past one another. The kinetic energy of the liquid particles is greater than for solid particles. A liquid flows and it can change shape, but it has a fixed volume. A liquid will take on the shape of its container, but not the volume.
The particles of a gas have very weak attractive forces, and their kinetic energy is very high. The particles of a gas are far apart. A gas has no fixed shape or volume — a gas will take on the shape and volume of its container.
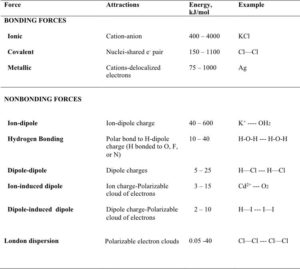
Intermolecular forces, IMFs, arise from the attraction between molecules with partial charges. The partial charges can also be induced. The following table compares both bonding and nonbonding forces. The energies of bonding forces are much higher than the energies of intermolecular forces (nonbonding forces). Note, that ion-induced dipoles and ion-dipole forces are not considered van der Waals forces. We will discuss these in a later study guide.
London Dispersion forces are lowest in energy, but the energy depends on the size of the molecule. The strengths of the dispersion forces increase with molar mass. Dipole-dipole forces are more energetic than London dispersion forces, and hydrogen bonding is an even stronger force of attraction between molecules.
Polarizability and Induced Dipoles
An electrical field can induce a distortion in the electron cloud of an atom, molecule, or an ion. For a nonpolar molecule, this will induce a temporary dipole moment. For a polar molecule, the electrical field will enhance the existing dipole moment. Smaller particles are less polarizable than larger ones because their electrons are held more tightly. Polarizability increases going down a group on the periodic table because of the increase in atomic size — the larger electron clouds are more easily distorted. Polarizability decreases across a period because of the increase in Zeff. Cations are less polarizable than their parent atoms because cations are smaller. Anions are larger than their parent atoms and are more polarizable.
London Dispersion Forces
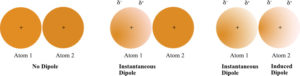
All molecules and atoms experience London dispersion forces of attraction. London Dispersion Forces are the result of an instantaneous dipole in a molecule or atom that will induce a temporary dipole in another particle resulting in an attraction between them. For example, Cl2 is a nonpolar molecule. Electrons are in motion and at any instant there can be a greater density of electrons on one side of the molecule resulting in an instantaneous dipole. The instantaneous dipole can induce an instantaneous dipole in another molecule and so on. This process will occur throughout a sample.
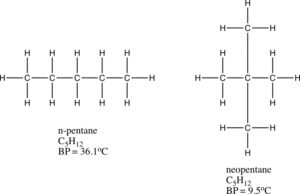
All molecules, whether polar or nonpolar experience these London dispersion forces. Atoms that are far apart will not influence one another. The strengths of the London dispersion forces increase with increasing polarity. The higher the molar mass of an atom or molecule, the stronger the dispersion forces of attraction. For example, HI has stronger dispersion forces than HF. The HI has a higher molecular mass. Molecular structure also influences the strength of dispersion forces. For example, n-pentane and neopentane have the same molecular formula and molar mass, but their structures are different. The n-pentane is a straight chain hydrocarbon, and the molecules are able to get closer to one another. In neopentane, the molecules have a branch and cannot get as close as the molecules in n-pentane. The n-pentane has the stronger dispersion forces. In fact, the boiling point of n-pentane is 36.1 °C which is higher than the boiling point of neopentane which is 9.5°C. The stronger dispersion forces between n-pentane molecules give rise to the higher boiling point.
Dipole-dipole Forces
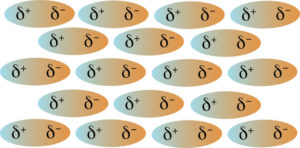
All polar molecules will experience dipole-dipole forces of attraction. The forces are due to the dipole of one molecule being attracted to the dipole of another molecule. Recall, a polar molecule has both a partial negative and a partial positive charge. The partial positive charge of one molecule is attracted to the partial negative charge of another molecule. This is shown in the figure below.
Acetone is a polar molecule with a molecular mass of 58.1 g/mol, and butane is a nonpolar molecule with a molar mass of 58.1 g/mol. Butane only experiences London dispersion forces of attractions where acetone experiences both London dispersion forces and dipole-dipole forces. Acetone has a boiling point of 58°C while butane has a boiling point of 30°C. The higher boiling point of acetone is due to the London dispersion forces and the dipole-dipole forces of attraction between the polar molecules. In many cases the London dispersion forces are stronger than the dipole-dipole forces in a molecule. This is true for larger molecules.
Hydrogen bonding
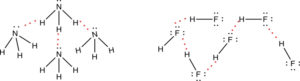
Hydrogen bonding is a special type of dipole-dipole intermolecular force. Hydrogen bonding is an attractive force between a hydrogen atom bonded to a very electronegative atom, O, N, or F, and an unshared electron pair on another O, N, or F atom. Hydrogen bonding is a very important intermolecular force in proteins and DNA. Water is a polar molecule and it experiences London dispersion and dipole-dipole forces. Water also experiences hydrogen bonding because there is a hydrogen bonded to an oxygen. The oxygen pulls the electron density of the bond toward itself, resulting in a partial positive charge on the hydrogen atom (more like a proton) and a partial negative charge on the oxygen atom. The partial charges are enhanced because of hydrogen bonding.
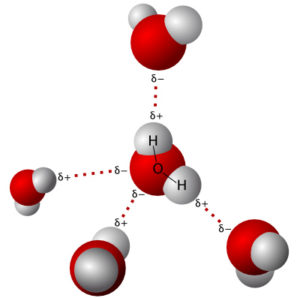
“File:3D model hydrogen bonds in water.svg” by User Qwerter at Czech wikipedia: Qwerter. Transferred from cs.wikipedia to Commons by sevela.p. Translated to english by by Michal Maňas (User:snek01). Vectorized by Magasjukur2 is licensed with CC BY-SA 3.0. To view a copy of this license, visit https://creativecommons.org/licenses/by-sa/3.0
Both NH3 and HF also experience intermolecular hydrogen bonding as shown below.
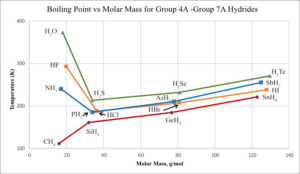
Below is a plot of the boiling points of the hydrides in Groups 4A – 7A. The boiling points of the compounds increase as we go down Group 4A due to the polarization, or dispersion forces, increasing with molar mass. Note, the same trend is followed for Groups 5A, 6A, and 7A when looking at the last three heavier compounds. But, H2O, HF, and NH3, have larger boiling points. This is because these three compounds have hydrogen bonding which results in them being higher boiling compounds. In fact water has the highest boiling point, and in addition it has a very high heat of vaporization and a high specific heat. The intermolecular forces are very strong, and this is due to hydrogen bonding between the molecules.
Recall, hydrogen has no inner electrons and it is therefore very small in H2O and other substances that experience hydrogen bonding. This means the attraction between the hydrogen atom and a lone pair of electrons is very strong, because the hydrogen can get very close to the unshared electron pairs in O, N, and F.
Worksheet: Intermolecular Forces Part 1
Worksheet: Intermolecular Forces Part 2
Exercises
Exercise 1. Indicate the intermolecular forces experienced by each of the following molecules.
b) PCl5
c) C4H10
d) CH2F2
Check Solution/Answer to Exercise 1
Exercise 2. Indicate the intermolecular forces that are experienced by each of the following.
b) O2
c) CHCl3
d) H2Se
e) C10H22
Check Solution/Answer to Exercise 2
Exercise 3. Below are the boiling points of some substances.
methanol, CH3OH, (64.7°C)
decane, C10H22, (174°C)
1-decanol, C10H21OH, (231°C)
Explain why methanol has a boiling point that is 229 degrees higher than that of methane while 1-decanol boils only 57 degrees higher than decane.
Check Solution/Answer to Exercise 3
Exercise 4. Draw a sketch of three methanol, CH3OH, molecules hydrogen bonding.
Check Solution/Answer to Exercise 4
Exercise 5. Which substance in each pair has the highest boiling point?
b) NH3 or PH3
c) H2S or H2Te
d) NOF or NOCl
Check Solution/Answer to Exercise 5
Exercise 6. Identify the intermolecular forces for each of the following substances.
b) NH2OH, hydroxylamine
c) PH3O, phosphine oxide
Check Solution/Answer to Exercise 6
Back to Bonding Models for Covalent Compounds
Back to General Chemistry 1 Study Guides
Back to Home Page