Bond polarity has already been discussed. Now, we move on to molecular polarity. It is important to be able to determine if a molecule is polar or nonpolar. There are two main things to consider here. In order for a molecule to be polar, it must
2. have an asymmetrical geometry
The polarity is dependent on the geometry of the molecule and the polarities of the individual bonds. If a molecule has all nonpolar bonds, the molecule itself is nonpolar. If there are one or more polar bonds, the polarity will depend on the geometry of the molecule. A bond dipole is a vector — it has both magnitude and direction associated with it. The dipole moment of a molecule is a sum of the vector quantities. The dipole moment, μ of a molecule is the product of the magnitude of the charge, δ, and the distance, d, that separates the centers of positive and negative charge.
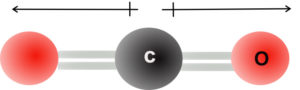
For example, for CO2, both C=O bonds are polar, but the molecule is nonpolar. This is because CO2 has a linear geometry as shown below.
The bond dipoles are equal and they pull in opposite directions which cancel out any dipole moment for the CO2 molecule. The molecule is nonpolar and the dipole moment is equal to 0.
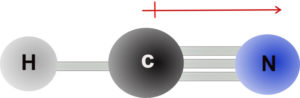
Below is the structure of HCN which is also linear. The C-H bond is nonpolar, but the C≡N bond is polar. In this case, the bond dipole moments do not cancel and they are summed to give a dipole moment of 2.98 Debye.
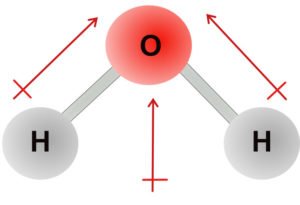
Water has a bent molecular geometry. The H-O bonds are polar, but because of the bent shape, the dipoles do not cancel, and the dipoles sum to give an overall dipole moment of 1.84 Debye. The water molecule is polar.
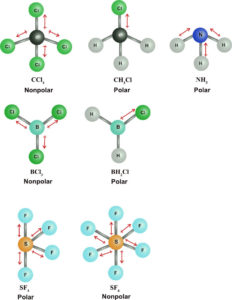
Below are some structures of other molecules that have one or more polar bonds. In general if you have a tetrahedral molecular geometry, AX4, with identical polar bonds, the molecule is nonpolar. On the other hand, if one bond is polar and the other three are nonpolar, you have a polar molecule. A trigonal planar geometry, AX3, will be polar if only one or two of the bonds are polar. If all bonds are polar, the molecule is nonpolar. Symmetry dictates whether or not a molecule is polar if there are one or more polar bonds in the molecule.
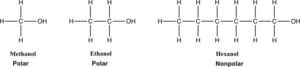
Many organic compounds, compounds that contain carbon, have polar groups. Usually if there are more than 4 carbon atoms, the molecule is considered nonpolar. For example, methanol and ethanol are polar, but the 6 carbon hexanol is much less polar. Because of its longer hydrocarbon chain, it has the characteristics of a hydrocarbon. The London dispersion forces of attraction are more important. Only a minuscule amount of hexanol will dissolve in water.
In order for a molecule to dissolve in water, it must be polar. Likes dissolve likes, therefore polar solvents will dissolve polar solutes, and nonpolar solvents will dissolve nonpolar solutes. Oil is a nonpolar compound, and will not dissolve in a polar compound like water. Oil will dissolve in nonpolar compounds. Ions are not considered polar or nonpolar — they are charged species and not molecules. Many ionic compounds dissolve in water and some other polar substances. We will discuss solubility and solutions in another study guide.
Exercises
Exercise 1. Indicate if SiCl4 and SCl4 are polar or nonpolar.
Exercise 2. Explain why CO2 has a zero dipole moment while SO2 has a dipole moment of 1.63 D.
Exercise 3. Which of the following would have a nonzero dipole moment? Show the dipole direction for each polar molecule.
b) PCl3
c) XeF2
d) BF3
Exercise 4. Does H2C=CHCl have a dipole moment? If so, draw the direction of the dipole moment.
Check Answers/Solutions to Exercises
Back to Bonding Models for Covalent Compounds
Back to General Chemistry 1 Study Guides
Back to Home Page