Answers/Solutions to Exercises
Exercises
Exercise 1. Indicate the intermolecular forces experienced by each of the following molecules.
This is a polar molecule.
It experiences both London dispersion forces and dipole-dipole forces of attraction.
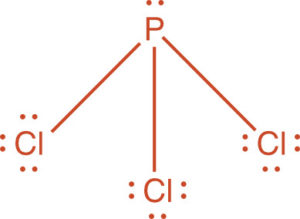
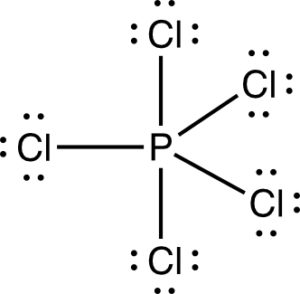
b) PCl5
This is a non polar molecule.
It only experiences London dispersion forces.
c) C4H10
This is a nonpolar molecule.
It only experiences London dispersion forces.
d) CH2F2
This molecule is polar.
It experiences both London dispersion forces and dipole-dipole forces of attraction.
Exercise 2. Indicate the intermolecular forces that are experienced by each of the following.
This molecule is polar.
It experiences both London dispersion and dipole-dipole forces of attraction. In addition, hydrogen is bonded to oxygen, and it experiences hydrogen bonding.
b) O2
O2 is nonpolar. It only experiences London dispersion forces
c) CHCl3
This molecule is polar. It experiences London dispersion and dipole-dipole forces of attraction.
This molecule is polar. It has a bent shape. It experiences both London dispersion and dipole-dipole attractive forces.
e) C10H22
This molecule is nonpolar. It only experiences London dispersion forces.
Exercise 3. Below are the boiling points of some substances.
methanol, CH3OH, (64.7°C)
decane, C10H22, (174°C)
1-decanol, C10H21OH, (231°C)
Explain why methanol has a boiling point that is 229 degrees higher than that of methane while 1-decanol boils only 57 degrees higher than decane.
Both methane and methanol have small dispersion forces of attraction. Methanol can hydrogen bond which explains the much higher boiling point.
Both 1-decanol and decane have large similar dispersion forces. The 1-decanol can also hydrogen bond, which is why it has a 57 degree higher boiling point than decane. The London dispersion forces are much stronger in decane and decanol than in methane and methanol.
Exercise 4. Draw a sketch of the hydrogen bonding in several molecules of methanol, CH3OH.
Exercise 5. Which substance in each pair has the highest boiling point?
S2Cl2 has the highest boiling point. Both molecules have a bent molecular geometry about the sulfur atoms, but Cl is larger than F, therefore, the dispersion forces are greater in S2Cl2 than in S2F2.
b) NH3 or PH3
Both molecules are polar, but NH3 also has intermolecular hydrogen bonding. NH3 has the highest boiling point.
c) H2S or H2Te
H2Te has the higher boiling point. Both molecules are polar, have a bent molecular geometry, and experience dipole-dipole forces. They both experience London dispersion forces. H2Te has the higher boiling point because Te is heavier than S and therefore H2Te experiences stronger dispersion forces.
d) NOF or NOCl
Both molecules have London dispersion forces and dipole-dipole attractive forces. Their molecular geometry is trigonal pyramidal. The NOCl has a higher boiling point because Cl is heavier than F, and therefore the dispersion forces of attraction are stronger in NOCl.
Exercise 6. Identify the intermolecular forces for each of the following substances.
This molecule experiences London dispersion, dipole-dipole, and hydrogen bonding forces of attraction. The hydrogen bonding forces are especially strong since there are two hydroxyl groups.
b) NH2OH, hydroxylamine
This molecule experiences London dispersion, dipole-dipole, and hydrogen bonding forces of attraction.
c) PH3O, phosphine oxide
The molecule is polar and experiences both London dispersion forces and dipole-dipole forces.