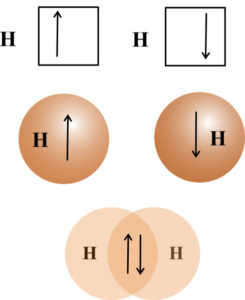
According to valence bond theory covalent bonds are formed by the overlap of atomic orbitals. The two overlapping lobes must be in the same phase. Each bonded atom maintains its own atomic orbitals, but the electron pair in the overlapping orbitals is shared by both atoms. The greater the orbital overlap, the stronger the bond. Because the strength of bonding is dependent on the orbital overlap, orbitals other than s orbitals bond only in given directions. The s orbitals are spherical and do not have directional lobes. Overlap of two s orbitals of two hydrogen atoms will result in a covalent bond. One valence electron and one valence orbital is provided by each hydrogen atom. Once the bond is formed the electron density resides between the two hydrogen nuclei.
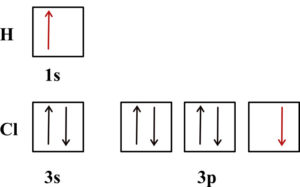
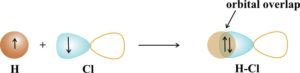
The hydrogen chloride bond, HCl, is formed from the overlap of a 1s orbital of hydrogen with chlorine’s 3p orbital. The 1s orbital of hydrogen has an unpaired electron and one of the 3p orbitals of chlorine has an unpaired electron. These are the orbitals that will overlap to form the H-Cl bond.
The p orbitals bond in the direction in which they point to obtain maximum overlap. For HCl, the bond is oriented along the axis of the p orbital on chlorine.
Diatomic chlorine, Cl2, has a covalent bond formed by the overlap of two 3p orbitals. The overlap of the 3p orbitals is end-to-end, and it provides a strong covalent bond. The maximum overlap is achieved with end to end overlap of p orbitals. Later, we will discuss side-to-side overlap of p orbitals.
Note, orbitals are mathematical functions. The p orbitals have two lobes and one of the lobes is shaded and the other is not. This is because the lobes have opposite signs in magnitude. The lobes that overlap must be in the same phase in order for a bond to form.
The bonds that we have discussed up to now are called sigma (σ) bonds. All single bonds are sigma bonds. If asked how many sigma bonds in C2H6, we would draw the structure and then count all of the single bonds. There are 7 sigma bonds in C2H6.
Some important points of valence bond theory are:
The bonding electrons are localized in the region of atomic orbital overlap.
For orbitals with directional lobes, the maximum overlap occurs when the atomic orbitals overlap end-to-end. This means a hypothetical line that joins the nuclei of the bonded atoms passes through the region of maximum overlap.
In the next study guide, we will discuss hybridization schemes.
Worksheet: Valence Bond Theory and Hybridization
Back to Bonding Models for Covalent Compounds
Back to General Chemistry 1 Study Guides
Back to Home Page