Exercises
Exercise 1. Balance the following chemical equations.
a) Mg + HNO3 → H2 + Mg(NO3)2
b) P4O10 + H2O → H3PO4
c) C6H14 + O2 → CO2 + H2O
d) CaCN2 + 3 H2O → CaCO3 + NH3
e) SiCl4 + H2O → SiO2 + HCl
Exercise 2. Ammonia gas and molecular oxygen gas react to produce nitrogen monoxide gas and liquid water.
Write a balanced chemical equation.
Exercise 3. Consider the following chemical equation and interpret it in words (Make sure the equation is balanced).
Fe2O3 (s) + C (s) → Fe (s) + CO2 (g)
The balanced equation is: 2 Fe2O3 (s) + 3 C (s) → 4 Fe (s) + 3 CO2 (g)
There are 2 moles of iron(III) oxide that react with 3 moles of solid carbon to produce 4 moles of solid iron and 3 moles of carbon dioxide gas.
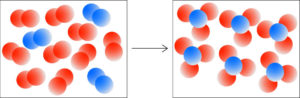
Exercise 4. Element A (red spheres) reacts with Element B (blue spheres). Write a balanced chemical equation for this process.
Count the number of reactant molecules and product molecules. The reactant box contains 9 red molecules, A2 and 3 blue molecules, B2. The product box contains 6 A3B molecules.
The reaction is: 9 A2 + 3 B2 → 6 A3B
We can divide the coefficients in the equation by 3 and our balanced equation is:
3 A2 + B2 → 2 A3B