Chemical formulas tell us the number of each type of atom in a compound. The two main types of compounds are covalent and ionic compounds. Recall, covalent compounds are composed of atoms that are covalently bonded through the sharing of electrons. Examples of covalent compounds are CO2, HCl, and CH4. In ionic compounds, electrons are transferred from the cation to the anion. Examples of ionic compounds are NaCl, Fe(OH)2, and NH4NO3. The smallest unit of a covalent compound is the molecule. The smallest unit of an ionic compound is the formula unit.
We will learn in a later study guide that ionic compounds that are water soluble will dissociate into ions when dissolved in water. This means that 1 mole of NaCl will dissociate into 2 moles of ions–1 mole Na+ and 1 mole Cl– ions. Most covalent compounds do not dissociate into ions when dissolved in water, but exceptions are strong and weak acids. These will be discussed later. For now, we will focus on atomic mass and how to calculate the atomic mass of an element or a compound.
Atomic mass is defined as the weighted average masses of the naturally occurring isotopes of an element. In a previous study guide, we defined the units of atomic mass as amu, atomic mass units (can also be represented by u). One amu is defined as 1/12 of a Carbon-12 atom.
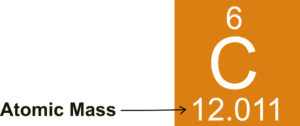
The atomic masses for all of the elements can be found on the periodic table. From the figure below, carbon has an atomic mass of 12.011 amu.
Atomic masses allow us to measure large amounts of atoms by weighing a sample of a compound or element. In fact, we can measure the number of carbon atoms required to draw this charcoal sketch of my beautiful Layla and Oliver. 
We know the atomic mass of carbon is 12.011 amu. The mass of the paper was 4.53 g and the mass of the paper with the charcoal drawing is 4.55 g. How many carbon atoms were used for the drawing?
The mass of charcoal used for the drawing is 4.55 g – 4.53 g = 0.02 g of charcoal
It took 1 x 1021 carbon atoms to draw the picture. There is another way we could have worked the problem, but first the SI quantity for amount, the mole, has to be discussed. One mole is equivalent to 6.02 x 1023 entities. This is referred to as Avogadro’s Number, NA. By entities, we mean atoms, molecules, ions, formula units, etc. For example, a carbon atom has an atomic mass of 12.011 amu. One mole of carbon atoms has a molar mass, Mm, equal to 12.011 g. The molar mass of an element is equal to its atomic mass in g/mol. The molar mass of sodium is 22.9898 g/mol. The molar mass can be used as a conversion factor to convert between grams and moles or vice versa. It can also be used to determine the number of atoms or molecules in a compound. Below are several examples.
Example 1. How many grams of nitrogen are in 5.45 moles of nitrogen?
Nitrogen has a molar mass of 14.0067 g/mole. When converting the number of moles to grams, multiply the moles by the molar mass:
Example 2. How many moles of copper are in 232 g of copper?
Copper has a molar mass of 63.546 g/mole. When converting from grams to moles, divide by the molar mass:
Example 3. How many iron atoms are in 245 g of iron? The first thing we do is look up the molar mass of iron which is 55.845 g/mol. We are converting grams of iron to atoms. We need a roadmap.
Two conversion factors are required. First we need to get from grams to moles and then from moles to grams. The molar mass of iron is equal to 55.845 g and 1 mole of iron is equivalent to 6.02 x 1023 iron atoms. Following the road map, use the molar mass to convert grams of iron to moles of iron and then convert moles of iron to the number of atoms of iron.
Sodium chloride, NaCl is an ionic compound with a molar mass of 58.44 g/mol. To calculate the molar mass of a compound, add the molar masses of each element in the compound. For NaCl the molar mass is calculated as:
For the covalent compound, glucose, C6H12O6, the molar mass of carbon is 12.011 g/mol, hydrogen is 1.00794 g/mol, and oxygen is 15.9994 g/mol. The molar mass for each element must be multiplied by the subscript in the chemical formula.
The molar mass of glucose is 180.158 g/mol. It would also be correct to state the molecular mass of glucose is 180.158 g/mol. Only use the term molecular mass for covalent compounds. Ionic compounds are not molecules–referring to their molar mass as molecular mass would be incorrect. The term formula mass can be used for ionic compounds as well as covalent compounds, but it is more commonly used for ionic compounds.
To convert grams to moles, divide the number of grams of compound by the molar mass of the compound. For example, to convert 32.5 g of NaCl to moles, the molar mass of NaCl must be calculated. The molar mass of NaCl is 58.443 g/mole (see above for the calculation). Next, divide the 32.5 g of NaCl by the molar mass:
To convert 3.50 moles of MgCl2 to moles, multiply the 3.50 moles by the molar mass of MgCl2.
If we ask how many moles of chlorine are in 1 mole of MgCl2, the correct answer would be 2 moles of chlorine. There is 1 mole of Mg in one mole of MgCl2.
One mole of caffeine, C8H10N4O2 has 8 moles of carbon, 10 moles of hydrogen, 4 moles of nitrogen, and 2 moles of oxygen. Two moles of caffeine would have 16 moles of C, 20 moles H, 8 moles N, and 4 moles O. How many moles of nitrogen are in 255 grams of caffeine? First, calculate the molar mass of caffeine and then convert to moles. Use the formula subscript for nitrogen to determine the number of moles in 255 g of caffeine.
Mm = 8 x 12.011 g/mol + 10 x 1.00794 g/mol + 4 x 14.0067 g/mol + 2 x 15.9994 g/mol = 194.193 g/mol
Worksheet: The Mole
Worksheet: Molecular Mass, Molar Mass, and Formula Mass
Worksheet: Atomic Mass, Number of Atoms and the Mole
Please watch the following videos before attempting exercises
Exercises
Exercise 1. How many moles of phosphorous are in 2.78 x 1024 atoms of phosphorous?
View Answer/Solution to Exercise 1
Exercise 2. How many grams of chlorine in 32.5 moles of chlorine?
View Answer/Solution to Exercise 2
Exercise 3. How many selenium atoms are in 235 grams of selenium?
View Answer/Solution to Exercise 3
Exercise 4. Calculate the molar mass of (NH4)3PO4.
View Answer/Solution to Exercise 4
Exercise 5. What is the molecular mass of table sugar (also called sucrose), C12H22O11?
View Answer/Solution to Exercise 5
Exercise 6. How many moles of calcium ions are in 625 g of Ca3(PO4)2? How many phosphate ions are in 625 g of Ca3(PO4)2?
View Answer/Solution to Exercise 6
Exercise 7. Calculate the number of grams of calcium in 3.45 x 1023 formula units of Ca(NO3)2.
View Answer/Solution to Exercise 7