In the Reactions in Aqueous Solutions study guide, we talk about reactions that take place in water. In this section, we will discuss how substances behave when placed in water. We know that table sugar, sucrose (C12H22O11) and NaCl, salt, both dissolve in water. When NaCl dissolves in water, it dissociates into Na+ and Cl– ions. Each of the ions are surrounded by water molecules. Recall, NaCl is an ionic compound. When a substance dissociates into ions when dissolved in water, the solution is able to conduct an electrical current. Each of the ions, Na+ and Cl–, is surrounded by water molecules.
Sucrose will dissolve in water as neutral sucrose, (C12H22O11) molecules. The neutral molecules are surrounded by water molecules.
Substances that do not dissociate into ions when dissolved in water are non-electrolytes. Some examples of non-electrolytes are alcohols and sugars. Substances, like ionic salts, that dissociate in water to produce ions are called electrolytes. Ionic salts like NaCl and CaCl2 that dissolve in water are strong electrolytes. Strong acids, like HCl, are also strong electrolytes because they essentially completely dissociate into ions in water. Weak acids, such as acetic acid (CH3COOH) do not completely dissociate into ions when placed in water. They only partially dissociate making them weak electrolytes. For example, only 1.3% of acetic acid in a 0.10 M aqueous solution dissociates. Compounds that dissociate to a great extent, about 70 to 100%, are strong electrolytes while compounds that dissociate to a smaller extent are weak electrolytes. Compounds that do not dissociate into ions, are non-electrolytes. Please see the table below.
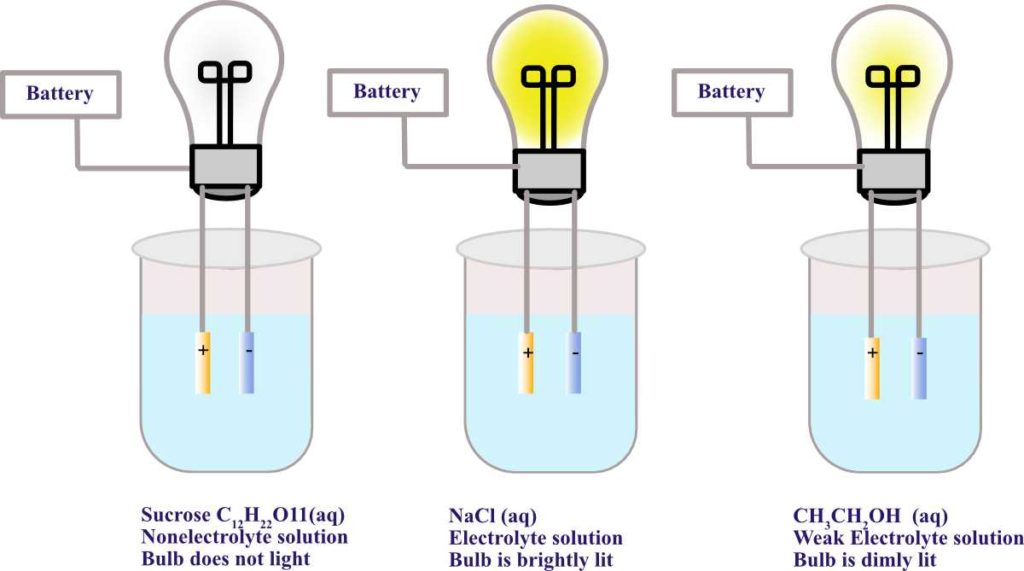
In the figure below, there is a lightbulb, a battery, and electrodes. When the electrodes are placed in the sucrose solution, the bulb does not light because sucrose is a non electrolyte–it does not dissociate into ions. The bulb lights when the electrodes are placed in the sodium chloride solution. There are ions in solution and the positive ions move toward the negative electrode and negative ions toward the positive electrode. The circuit is completed because of the presence of these ions in solution.
In the figure above, note the aqueous acetic acid solution. It can be seen the bulb does light up, but it is much dimmer than the light from the bulb with the aqueous ionic solution. This is because CH3COOH is a weak acid and therefore, a weak electrolyte. It only partially dissociates in water. In conclusion, the greater the number of ions in aqueous solution, the higher the electrical conductivity or the brighter the bulb.
Next, we look at concentrations of ions in aqueous solution. A strong electrolyte like Na2SO4 dissolves in water. What is the molar concentration of sodium and sulfate ions in the solution if we have a 0.375 M solution? Assume complete dissociation. First, write the equation for the dissociation.
From 1 mole of sodium sulfate, there are 2 moles of Na+ ions and 1 mole of SO42- ions in the aqueous solution. The solution is 0.375 M Na2SO4. This means there are
2 x 0.375 M Na+ ions = 0.750 M Na2SO4 ions
1 x 0.375 M SO42- ions = 0.375 M SO42- ions
The total molar concentration of ions in the solution is 0.750 M + 0.375 M = 1.125 M total ions.
The molar concentration of a nonelectrolyte solution is the same as given initially since there is no dissociation. As far as weak electrolytes go, we will learn a method later in General Chemistry 2 to determine concentrations. If a substance only partially dissociates, more information is required to solve the problem.
Exercises
Exercise 1. Is hydrofluoric acid, HF, a strong, weak, or nonelectrolyte?
Exercise 2. Indicate each of the following as a nonelectrolyte, weak electrolyte or strong electrolyte.
a) FeCl3
b) H2SO3
c) C6H12O6
d) CaClO4
e) CH3CH2OH
f) Na2CO3
Exercise 3. What is the molar concentration of Mg+2 ions in an aqueous solution that is 0.455 M in Mg3(PO4)2. What is the molar concentration of PO43-?
Exercise 4. What is the total molar concentration of ions in an aqueous 1.570 M K2CrO4?
Exercise 5. An aqueous solution is prepared by mixing 0.625 g of K2SO4, 1.264 g of K3PO4, and 0.543 g Na2SO4 and diluting with water to 250.00 mL. What is the molar concentration of each ion in the solution?
Back to Reactions in Aqueous Solution
Back to Study Guides for General Chemistry 1
Back to Home Page


I like the valuable information you provide in your articles.
I’ll bookmark your weblog and check again here regularly.
I’m quite certain I’ll learn a lot of new stuff right here!
Good luck for the next!