The third category of reaction to discuss are oxidation-reduction reactions. Many of the reactions around us are redox (short for oxidation-reduction) reactions. The rusting of metal, tarnishing of silver, and many biological reactions are redox reactions. Before discussing these reactions a few definitions are in order. Oxidation is defined as the loss of electrons and reduction is the gain of electrons. The substance that is oxidized or reduced can be an element, a compound, or an ion. For many organic reactions, oxidation is an increase in the number of C–O bonds usually with a simultaneous decrease in the number of C–H bonds. Reduction is an increase in the number of C–H bonds with a simultaneous decrease in the number of C–O bonds.
To determine if we have a redox reaction, each atom in a substance is assigned an oxidation number also called an oxidation state. Oxidation numbers allow us to determine if a substance is electron rich, electron poor, or neutral. If the oxidation number of an atom changes in a reaction, we have a redox reaction. Below are the rules for assigning oxidation numbers.
1. Atoms in their elemental state have oxidation numbers of 0. Some examples are:
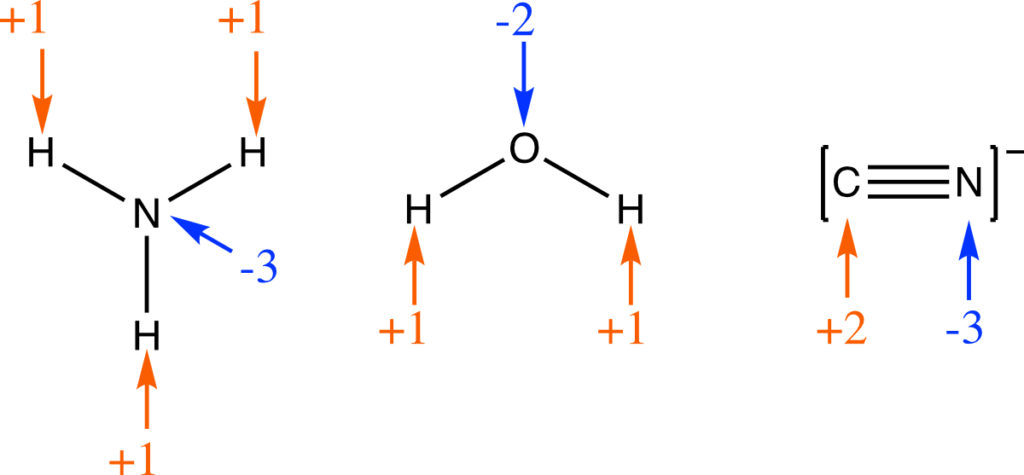
2. A monotomic ion has an oxidation number equal to the charge on the ion. Some examples:
![]()
3. An atom in a polyatomic ion or in a molecular compound will usually have the same oxidation number as if it were a monotomic ion.
4. Hydrogen usually has an oxidation number of +1 except when it is bonded to a metal in which case the oxidation number is -1.
5. In most compounds, oxygen has an oxidation number of -2. The main exception is in peroxides where the oxidation number is -1.
6. Halogens usually have an oxidation number of -1. Some exceptions are below.
6. The sum of oxidation numbers must add to the charge on the species.
Let’s do some examples.
2. What is the oxidation number of the atoms in CoCl2? The oxidation number of Cl is -1. There are two Cl, therefore the oxidation number of Co is +2. This is a binary ionic compound and the oxidation numbers are equal to the charges on each ion.
3. What is the oxidation number of the atoms in NO3–? The oxygen has an oxidation number of -2. The oxidation numbers must add to the -1 charge on the ion. We have 3 oxygens and one nitrogen. 3 x -2 + ? = -1 and ? = +5. The nitrogen has an oxidation number of +5. 3 x -2 + 5 = -1.
4. What is the oxidation number of sulfur in H2SO4? The oxidation number of each of the two hydrogens is +1 and the oxidation number of each of the 4 oxygens is -2. The oxidation numbers must sum to 0. We have: 2 x +1 + 4 x -2 + ? = 0 and ? = +6. There is one sulfur atom and its oxidation number is +6.
5. What is the oxidation number of Cr in K2Cr2O7? Each of the two potassiums has an oxidation number of +1 and and each of the 7 oxygens have an oxidation number of -2. We have: 2 x +1 + 7 x -2 + ? = 0 and ? = +12 There are two chromium atoms and we divide the 12 by 2 to get 6. The oxidation number of chromium is +6.
It is important that you learn how to assign oxidation numbers, so keep practicing.
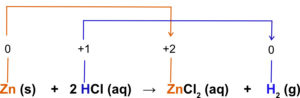
Now, we are ready to discuss oxidation-reduction or redox reactions. You might have noticed that when a substance is oxidized, the oxidation number increases. This corresponds to a loss of one or more electrons. When a substance is reduced, the oxidation number will decrease. This corresponds to a gain of one or more electrons. The reaction below is a redox reaction. How do we know this? Assign oxidation numbers for each of the atoms in the reaction. If an oxidation0-reduction has occurred, there will be a change in oxidation number. For a reaction, at least two oxidation numbers must change in order for the reaction to be a redox reaction.
The oxidation number of Zn changes from 0 to +2. The oxidation number has increased meaning the atom has lost electrons. The Zn has been oxidized. The oxidation number for the hydrogen in HCl has changed from +1 to zero for the hydrogen gas. The oxidation number has decreased meaning electrons have been gained and the H+ has been reduced.
The net ionic equation for the reaction is:
The solid zinc has been oxidized and the H+ has been reduced. For all redox reactions, if a substance is oxidized another substance must be reduced and vice versa. The substance that causes oxidation is the oxidizing agent. The substance that causes reduction is the reducing agent. In the above reaction, the Zn (s) is oxidized, therefore it is the reducing agent. The H+ ion is reduced, therefore it acts as the oxidizing agent. The substance that is oxidized always acts as a reducing agent and the substance that is reduced always acts as the oxidizing agent.
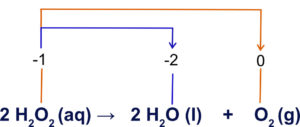
There are reactions, disproportionation reactions, where one substance can act as both a reducing agent and oxidizing agent. These are rare, but the decomposition of hydrogen peroxide is an example. Recall, the oxidation number of oxygen in a peroxide is -1.
We can see the oxygen is both oxidized and reduced in the reaction above.
Worksheet: Oxidation Reduction
The following videos might be helpful before attempting exercises.
Exercises
Exercise 1. Determine the oxidation number of nitrogen in the following compounds:
b) NO2
c) N2O5
d) N2O4
View Answers/Solutions to Exercise 1
Exercise 2. Determine the oxidation number of bromine in the following compounds and ions.
b) HBrO2
c) HBrO3
d) HBrO4
e) BrO4–
f) HBr
g) BrO2–
View Answers/Solutions to Exercise 2
Exercise 3. Answer the questions for the following reactions.
What is the oxidation number of Ag (s)?
Which substance is oxidized?
Which substance is reduced?
b) Cl2 (aq) + 2 NaI (aq) → I2 (aq) + 2 NaCl (aq)
Which substance is reduced?
Which substance is the oxidizing agent?
Which substance is the reducing agent?
c) CH3CH2OH (l) + 3 O2 (g) → 3 H2O (l) + 2 CO2 (g)
b) Which substance is the reducing agent?
c) Which substance is the oxidizing agent?
d) Which substance has been reduced?
View Answers/Solutions to Exercise 3
Exercise 4. Write a balanced equation for the reaction of solid magnesium with aqueous cobalt(II) sulfate to yield solid cobalt and aqueous magnesium sulfate. Indicate both the reducing agent and the oxidizing agent.
View Answers/Solutions to Exercise 4
Back to Reactions in Aqueous Solution
Back to General Chemistry 1 Study Guides
Back to Home Page