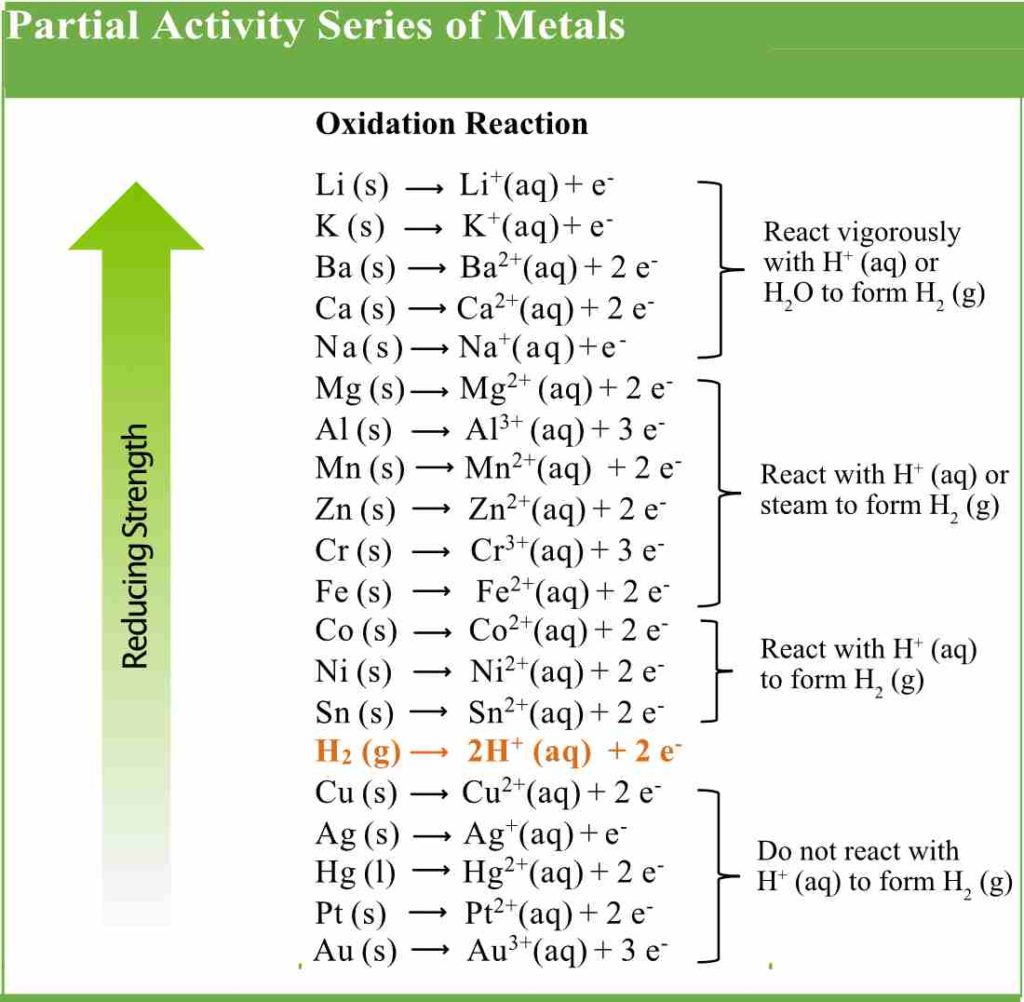
Metals are more easily oxidized than nonmetals. This means metals lose electrons more easily than nonmetals. Metals are strong reducing agents, but their reducing power decreases going across a period of the periodic table. Below is a table of a partial activity series for metals in aqueous solution. The table lists the oxidation reactions. Lithium is at the top of the list and is the most easily oxidized metal which means it is the strongest reducing agent in the activity series below. Gold is at the end of the list and is not as easily oxidized and is therefore the weakest reducing agent on this activity series.
Both the ease of oxidation and the reducing strength of the metals decrease going down the column. For example, zinc is a weaker reducing agent than sodium, but it is a stronger reducing agent than copper. A metal can reduce any ion below it in the series. Chromium can reduce Cu2+ ion, but it cannot reduce Mn2+.
Silver tarnishes according to the following reaction with H2S gas.
Commercial silver polishes will remove the tarnish from silver, but some of the silver is lost because these polishes are abrasive. Tarnish can be removed chemically with aluminum. The aluminum is a stronger reducing agent than silver and will reduce the Ag+ ion to solid Ag.
Notice the position of hydrogen in the activity series. The metals above hydrogen will react with aqueous hydrogen ion to form hydrogen gas. In fact, the first five metals in the series, both Group 1A and 2A metals, will react with pure water to form hydrogen gas.
The remaining metals (above hydrogen) will not react with water but do react with aqueous hydrogen ions. The metals below hydrogen in the series do not react with aqueous hydrogen ions or water. They are much more stable meaning they are less reactive. As we go from left to right on the periodic table, reactivity decreases.
Worksheet: The Activity Series
Exercises
Exercise 1. Consider the partial activity series below to answer the questions.
Mn(s) → Mn2+ (aq) + 2 e–
Fe(s) → Fe2+ (aq) + 2 e–
Ag(s) → Ag+ (aq) + e–
Hg(l) → Hg2+ (aq) + 2 e–
a) Which metal is the weakest reducing agent?
b) Will solid iron reduce manganese ion?
c) Will the following reaction occur? Fe(s) + Hg 2+ (aq) → Fe2+ (aq) + Hg (l)
Check Answers/Solutions to Exercise 1
Exercise 2. Use the partial activity series below to write a reaction with manganese and silver.
Mn(s) → Mn2+ (aq) + 2 e–
Fe(s) → Fe2+ (aq) + 2 e–
Ag(s) → Ag+ (aq) + e–
Hg(l) → Hg2+ (aq) + 2 e–
Check Answers/Solutions to Exercise 2
Exercise 3. Use the following activity series to write a balanced equation for the reactions below. If no reaction occurs, just write NR.
a) Fe (s) + Cu(NO3)2 (aq) →
b) Sn (s) + HBr (aq) →
c) Al (s) + CoSO4 (aq) →
d) Zn (s) + MgSO4 (aq) →
Check Answers/Solutions to Exercise 3
Exercise 4. Use the following reactions to arrange an activity series for the elements A, B, C, and D.
B + D+ → B+ + D
C+ + D → no reaction
B + C+ → B+ + C
Check Answers/Solutions to Exercise 4
Back to Reactions in Aqueous Solution
Back to General Chemistry 1 Study Guides
Back to Home Page