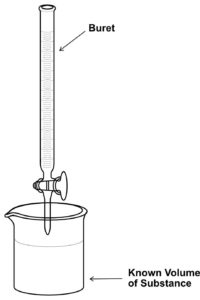
A titration is an analytical technique where a measured volume of a solution with an unknown concentration is combined with a second solution with known volume and known concentration and is called a standard solution. Just enough of the standard solution is added to completely react with the solute in the solution with the unknown concentration. The equivalence point is when stoichiometric amounts of reactants have reacted.
Titrations can be acid-base, oxidation-reduction, or precipitation reactions. Here the focus will be mainly on a neutralization reaction. A solution that contains a known volume of an unknown concentration of substance is placed in a beaker or an erlenmeyer flask. The titrant is the substance with a known concentration. The titrant is placed in a buret, and small volumes of titrant are added to the reaction flask until the substance has been completely reacted. We add a few drops of an indicator to the beaker which changes the color of the solution once all of the substance in the beaker has been reacted with the titrant. This is the endpoint of the titration.
Calculations for titrations are worked in the same way as the ones that were done in the Solution Stoichiometry study guide. Let’s do an example. What is the molar concentration of HNO3 if 25.00 mL of HNO3 was titrated with 0.105 M NaOH, and it took 21.65 mL of NaOH to reach the endpoint? First, write the balanced chemical equation:
The NaOH is the titrant and we can determine the concentration by multiplying the molarity of NaOH by the volume, in liters, that it took to reach the equivalence point.
\(\displaystyle moles\;NaOH\;=\;0.105\;mol/L\;\times\;0.02165\;L\;=\;\mathbf{0.00227\;mol\;NaOH} \)
From the balanced equation, there is a 1:1 mole ratio of NaOH to HNO3. At the equivalence point, 0.00227 mol of NaOH has reacted with 0.00227 mol of HNO3.
\(\displaystyle 0.00227\;mol\;NaOH\times\frac{1\;mol\;HNO_3}{1\;mol\;NaOH}\;=\;\mathbf{0.00227\;mol\;HNO_3} \)
Now, divide the number of moles of HNO3 by the volume, in liters, of the acid to calculate the molar concentration of HNO3.
We will discuss titrations in much more detail in the General Chemistry 2 Study Guides.
Worksheet: Volumetric Stoichiometry and Titration
Please watch the following videos before attempting the exercises.
Exercises
Exercise 1. A 50.00 mL aqueous HCl solution is titrated with NaOH. It takes 37.62 mL of 0.254 M NaOH to reach the endpoint. What is the concentration of HCl?
Check Solution/Answer to Exercise 1
Exercise 2. A 40.00 mL aqueous H2SO4 is titrated with KOH. It takes 18.35 mL of 0.325 M KOH to complete the reaction. What is the concentration of H2SO4?
Check Solution/Answer to Exercise 2
Exercise 3. Consider the following net ionic equation:
It takes 18.62 mL of 0.1625 M K2Cr2O7 to tirate a sample of FeSO4. What was the mass of the FeSO4 sample?
Check Solution/Answer to Exercise 3
Exercise 4. Consider the following equation:
How many mL of 0.245 M Na2S2O3 solution is needed to completely react with 2.252 g of I2?
Check Solution/Answer to Exercise 4
Exercise 5. The amount of iron in an ore can be determined by oxidizing the sample with Br2 to convert all of the iron to Fe3+. The Fe3+ is then titrated with Sn2+ to reduce the iron(III) ion to Fe3+ according to the following equation:
Calculate the mass percent of Fe metal in a 0.1725 g sample of ore if 11.65 mL of 0.09505 M Sn2+ is used to titrate Fe3+.
Check Solution/Answer to Exercise 5
Back to Reactions in Aqueous Solution
Back to General Chemistry 1 Study Guides
Back to Home Page