Solutions to Exercises
Exercise 1. Calculate q, if 55.26 g of water at 85.0 °C is cooled to 22.53°C? What is the sign of q? Cwater = 4.184 J/(g⋅K)
q = m x C x ΔT and ΔT = 22.53°C – 85.0°C = -62.5°C
\(q\;=\;55.26\;g\;\times\;4.184\frac{J}{g⋅K}\times\;-62.5°K\;=\;\mathbf{-14450\;J\;=\;-14.5\;kJ}\)
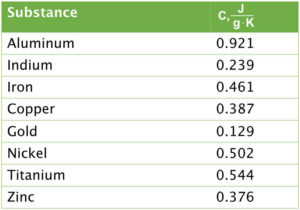
Exercise 2. An unknown metal has a mass of 15.65 g. The metal was heated to 190.5°C and added to 65.00 g of H2O with a temperature of 22.5°C. Once thermal equilibrium was established, the temperature of the H2O was 27.6°C. CH2O = 4.184 J/(g⋅K). Which of the metals in the table below could be the unknown metal?
The metal will release heat into the water.
qwater = -qwater
We need to find the specific heat capacity of the metal. First, find qwater
qwater = 65.00 g x 4.184 J/(g⋅°C) x (27.6°C – 22.5°C) = 1387 J
Because qwater = -qwater, qmetal = -1387 J
qmetal = mmetal x Cmetal x ΔT and \(\displaystyle C_{metal}\;=\;\frac{q_{metal}}{m_{metal}\times\;ΔT}\)
\(\displaystyle C_{metal}\;=\;\frac{-1387\;J}{15.65\;g\times\;(27.6\;-\;190.5)°C}\;=\;\frac{0.544\;J}{g⋅°C}\)
Checking the table, the metal could be titanium.
Exercise 3. Consider the reaction, at constant pressure, of 150.00 mL of 0.350 M HCl and 150.00 mL of 0.350 M KOH. The temperature of the solutions before mixing was 23.10°C. After the reaction occurred, the temperature of the solution was 25.78°C. What is the enthalpy change per mole (kJ/mol) of the neutralized acid? Is the reaction endothermic or exothermic? Assume solution densities to be 1.00 g/mL and the specific heat capacities are 4.184 J/(g⋅K).
The reaction is the system and the solution is the surroundings. The volume of solution is 300.00 mL. We use the density of water to convert volume to mass of solution.
\(\displaystyle 300.00\;mL\;\times\frac{1.00\;g}{mL}\;=\;300.00\;g\)
Next we find ΔT. ΔT = 25.78°C – 23.10°C = 2.60 °C = 2.60 K
qsolution = 300.00 g x 4.184 g/(g⋅K) x 2.60 K = 3263.5 J
Next, we find ΔH. Find the amount, in moles, of both reactants, HCl and KOH.
massHCl = 0.15000 L x 0.350 M = 0.175 mol HCl
massKOH = 0.15000 L x 0.350 M = 0.175 mol KOH
The HCl and NaOH completely react–there is no limiting reactant. We found qsolution equal to 3263.5 J. Because the heat absorbed by the solution was the heat released by the reaction:
qsolution = -qreaction
qreaction = -3263.5 J This is the enthalpy change when 0.175 mol of HCl is neutralized. We are asked for ΔH when 1 mol is neutralized
\(\displaystyle 1\;mol\;HCl\;\times\frac{-3263.5\;J}{0.175\;mol}\;=\;\mathbf{-18648.6\;J/mol\;=\;-18.6\;kJ/mol}\)
The reaction is exothermic.
Exercise 4. An instant hot pack provides heat because of the exothermic dissolution of a salt.
CaCl2 (s) + H2O (l) → Ca2+ (aq) + 2 Cl– (aq) + H2O (l) ΔH = -82.8 kJ
What is the final temperature of a squeezed hot pack that contains 18.5 g of CaCl2 that is dissolved in 125.0 mL of water? Assume the solution has a specific heat of 4.184 J/(g⋅°C). The initial temperature was 22.3°C and assume the density of the solution is 1.00 g/mL.
q = m x c x ΔT
The amount of heat released when 1 mole of CaCl2 is dissolved is -82.8 kJ. This is the same as 8.28 x 104 J/mole.
Determine the amount of heat for 18.5 g of CaCl2.
18.5 g of CaCl2 is equal to 0.167 mol CaCl2.
\(\displaystyle 0.167\;mol\;CaCl_2\times\;\frac{8.28\times\;10^4\;J}{1\;mol\;CaCl_2}\;=\;13827.6\;J\)
We are looking for the final temperature of the water once the solute is dissolved. The density of water is 1.00 g/mL, therefore, 125.0 mL of H2 is equivalent to 125.0 g of H2.
Solve, q = m x c x ΔT, for Tfinal
q = m x C x (Tfinal-Tinitial)
\(\displaystyle T_{final}\;=\;\frac{13827.6\;J}{\frac{4.184\;}{g⋅°C}\times\;125.00\;g}\;+\;22.3°C\;=\mathbf{48.7\;°C}\)
Exercise 5. A 1.798 g sample of phenol (C6H5OH) was burned in a bomb calorimeter. The heat capacity of the calorimeter is 11.76 kJ/°C. The temperature of the calorimeter and its contents increased from 21.47 °C to 26.46°C. What is the heat of combustion for one mole of phenol in kJ/mol? (Specific Heat Capacity of H2O = 4.184 J/(g⋅°C)
qrxn = -qH2O – qcalorimeter
Here we are given the heat capacity, 11.76 kJ/°C. The calorimeter includes the bomb and the water. We need to write the balanced equation and then find ΔT.
2 C6H5OH (s) + 14 O2 (g) → 12 CO2 (g) + 6 H2O (g)
ΔT = 26.46°C – 21.47°C = 4.99°C
qreaction = 11760 J/°C x 4.99°C = 58682.4 J = 58.7 kJ
58.7 kJ of heat is released from the reaction when 1.798 g of phenol is burned. We need the amount of heat released when 1 mole (0.0191 moles) of phenol is burned.
\(\displaystyle 1.00\;mol\;\times\frac{58.7\;kJ}{0.0191\;mol}\;=\;\mathbf{3073\;kJ}\)