Many processes and chemical reactions are exothermic and occur spontaneously. But, there are also processes and reactions that are endothermic and occur spontaneously. For example, an ice cube will spontaneously melt at temperatures just above 0°C, but ice will not spontaneously melt at temperatures below 0°C. Melting of a solid is an endothermic process, and NH4NO3 will spontaneously dissolve in water in an endothermic process. A spontaneous process or reaction is one that does not require any external influence once started. A nonspontaneous process or reaction is one that does require a continuous external influence, and once that external influence is removed, the process or reaction will stop. An external influence is energy supplied by the surroundings.
Photosynthesis is an endothermic reaction. Plants absorb thermal energy from the sun to convert CO2 and H2O into glucose, C6H12O6, and O2.
Some spontaneous exothermic processes include freezing and condensation at low temperatures, combustion reactions, and oxidation of iron and other metals. Spontaneous endothermic processes include melting and vaporization at higher temperatures and the dissolving of many soluble salts.
It is obvious that enthalpy is not the only factor that predicts if a reaction or process is spontaneous. In fact, entropy is a thermodynamic quantity that measures or describes the molecular randomness of a system. A system of gas particles has a higher entropy than the particles of a liquid, and the particles in a liquid have a higher entropy than the particles of a solid. Entropy is represented by a capital S and has units of J/K.
Both enthalpy and entropy determine whether or not a process or reaction is spontaneous. Any spontaneous process or reaction that is endothermic, will show an increase in the molecular randomness of the system — an increase in entropy.
The change in entropy is given by:
Entropy is a state function which is only dependent on the initial and final states of a system and not on the path taken to get there. We can measure absolute entropies, and those are listed in the Table of Thermodynamic Quantities for different substances. Note, the entropy for an element in its most stable state is not zero. We will go into more detail about entropy in the General Chemistry II Study Guides.
The Gibbs free-energy change, ΔG, determines if a reaction or process is spontaneous. It is given by:
where T is the temperature in Kelvin. If ΔG is less than zero, the process or reaction is spontaneous. If ΔG is greater than zero, the process or reaction is nonspontaneous. When ΔG is equal to zero, the system is at equilibrium and the process is neither spontaneous or nonspontaneous. Equilibrium is when the forward and reverse rates of the process or reaction are equal.
A favorable ΔH is less than zero (ΔH < 0), and a favorable ΔS is greater than zero, ΔS > 0. If both ΔH and ΔS are favorable, a reaction or process is always spontaneous regardless of the temperature. If both ΔH and ΔS are unfavorable, ΔH > 0 and ΔS < 0, then a process or reaction is always nonspontaneous, regardless of temperature. If ΔH is greater than zero and ΔS is greater than zero, then the process or reaction is spontaneous at higher temperatures and nonspontaneous at lower temperatures. The temperature depends on the substance under study. If ΔH is less than zero and ΔS is less than zero, the reaction or process will be spontaneous at lower temperatures and nonspontaneous at higher temperatures. The table below summarizes this:
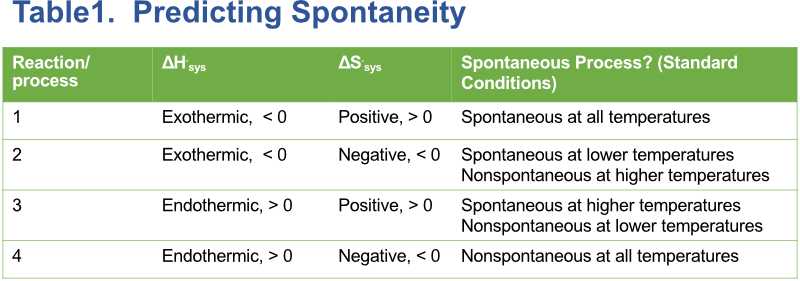
We see that temperature controls spontaneity if only one of the factors is favorable, either ΔH or ΔS, and the other factor unfavorable. Think of the solidification (freezing) of water. In this example, ΔH is favorable, but ΔS is unfavorable because the particles are becoming more ordered (less freedom of motion) as the liquid is converted to a solid, ΔS < 0. Freezing of water occurs at 0°C and below. At temperatures just above 0°C, the water will not freeze. At exactly 0°C, there is an equilibrium between liquid and solid, and the process is neither spontaneous or nonspontaneous. At equilibrium, ΔG is equal to zero. This means the process or reaction is at equilibrium and is neither spontaneous or nonspontaneous. If we set ΔG = 0;
Solving for T;
This equation allows us to calculate the temperature in which a change in spontaneous and nonspontaneous behavior occurs if ΔH and ΔS are known. For example, for the vaporization of water, ΔH° and ΔS° are 40.7 kJ/mol and 109 J/(K⋅mol), respectively.
For the vaporization of water, the process is nonspontaneous below 100°C and is spontaneous at temperatures above 100°C. At 100°C, the system is at equilibrium and the boiling point of water is 100°C. Both liquid and gas are in equilibrium.
Exercises
Exercise 1. Indicate if entropy is positive or negative for the following:
b) condensation
c) melting
Exercise 2. Indicate if entropy is increasing or decreasing in the following:
b) C2H4 (g) + Cl2 (g) → C2H4Cl2 (l)
c) Cl2 (g) + H2C=CH2 (g) → ClCH2CH2Cl (l)
Exercise 3. Indicate if reactions with the following ΔH and ΔS values are spontaneous, nonspontaneous, endothermic, or exothermic.
b) ΔH = -126 kJ; ΔS = 38 J/K at 485 K
c) ΔH = 78 kJ; ΔS = 98 J/K at 785 K
d) ΔH = 64 kJ; ΔS = -135 J/K
Exercise 4.
A process has ΔH = 32 kJ and ΔS = 56 J/K. At what temperature will it change from nonspontaneous to spontaneous?
Exercise 5.
Consider the following reaction:
Calculate ΔG° for the reaction at 25°C.
Check Answers/Solutions to Exercises
Back to Thermochemistry
Back to General Chemistry 1 Study Guides
Back to Home Page